Solutions
Advancing Bioprocessing
With a culture of innovation, and focused on cost and process efficiencies, Repligen specializes in applications of technologies and solutions that help set new standards in perfusion, process intensification, enhanced vector titer and recovery yield, continuous manufacturing, and more.

Driving Progress in Bioprocessing
Since 1981, Repligen has been a trusted partner to biopharmaceutical manufacturers, driving progress through innovative solutions that challenge the status quo. We're committed to redefining what's possible in bioprocessing, with cutting-edge technologies designed to meet your needs at every stage of biologic drug production.
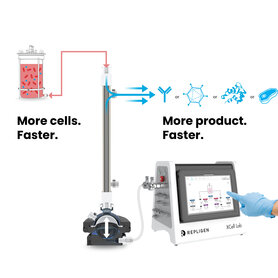
Process Intensification
MORE. FASTER.
Bioprocess intensification is defined as a significant step increase in output relative to cell concentration, time, reactor volume or cost, resulting in improvements in productivity, environmental and economic metrics.

Ultrafiltration | Diafiltration
Used in nearly every bioprocessing workflow, ultrafiltration/diafiltration (UF/DF) is a critical purification process, using tangential flow filtration (TFF), prior to final drug formulation.

Continuous Manufacturing
FROM BATCH TO CONTINUOUS
Continuous manufacturing is a method for manufacturing pharmaceutical products from end-to-end on a single, uninterrupted production line. The aim is to eliminate batch inefficiencies that result in process stoppages, long hold times, and sub-optimal facility utilization.

Real-time Process Management Solutions
Our Real-time Process Management (RPM™) Systems establish a new benchmark for real-time process control in downstream bioprocessing by integrating of the PATsmart™ FlowVPX® in-line UV-Vis spectrophotometer into the KrosFlo® TFF Systems. This innovative solution empowers users to achieve unparalleled control and optimization of UF/DF processes, driving efficiency, quality, and innovation in biomanufacturing.

Process Analytics
Our process analytics solutions, featuring Variable Pathlength Spectroscopy for at-line and in-line concentration monitoring systems, ensure that products meet required Process Analytical Technology (PAT) standards. These tools provide critical data for optimizing bioprocesses and ensuring compliance with regulatory requirements.

ATMP
Advanced Therapy Medical Products (ATMP) have immense potential to transform the lives of patients with genetic diseases. Repligen partners with developers and manufacturers, providing technology breakthroughs and high impact solutions for plasmid and viral vector development and manufacturing.

Fluid Management
Repligen offers innovative, designed-for-purpose storage and transfer solutions for fluid management within and between unit operations. These include flow paths, tubing, valves and gaskets, bottles and containers, totes, carts and cleanroom process equipment.