products
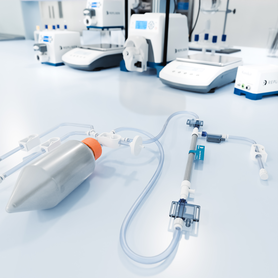
TangenX® SIUS® Gamma TFF Devices
With the performance and efficiency of SIUS® Cassettes, SIUS Gamma Devices come as a convenient, fully assembled, closed, and sterile system.
product features
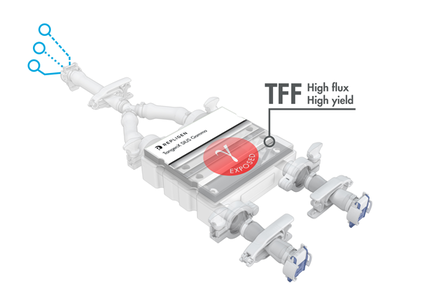
Achieve higher flux, closed device and connectivity freedom
TangenX® SIUS® Gamma TFF Devices embody the performance and efficiency of SIUS® Cassettes in a convenient, fully assembled, closed, and sterile system. Acclaimed performance with up to 30% flux increase over conventional membranes, connectivity freedom, and a library of configurations, converge into one, simple TFF solution. SIUS Gamma TFF Devices are ideal for gene therapy applications, bioburden-sensitive operations, and hazardous processes.
Ready-to-use
Clamp-and-go connectivity
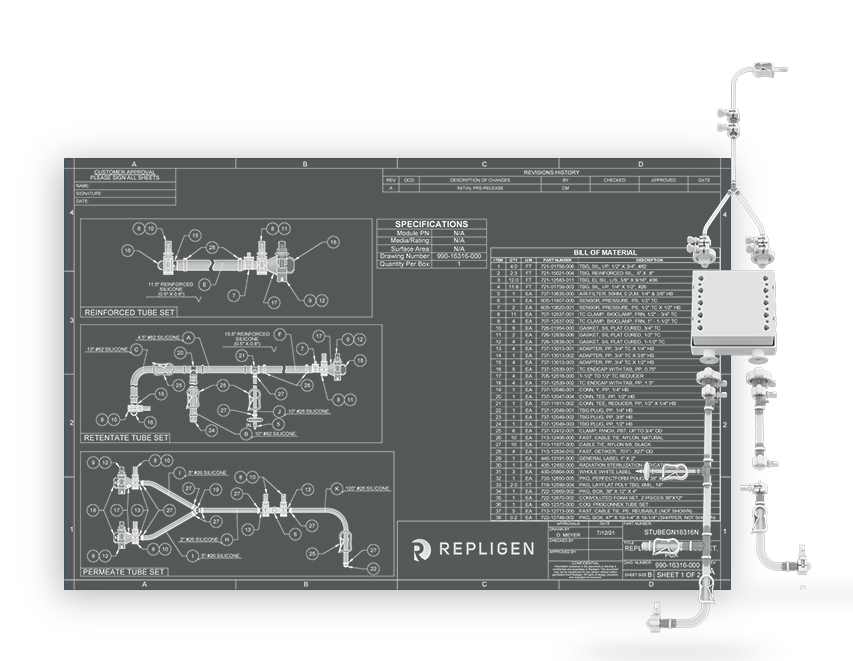
The diversity of connectivity options makes building closed systems a challenge. Genderless AseptiQuik® connectors featured on TangenX® SIUS® Gamma TFF Devices make life easier. Simply clamp the SIUS® Gamma TFF Device into a holder, connect to a flow path, and go. It's that easy.
Click on the links below to explore how to assemble SIUS Gamma TFF Devices. TangenX® SIUS® hardware may be purchased separately.
ready-to-use
Simple connectivity with ProConnex® Flow Paths
Custom ProConnex® Flow Paths complete your TFF solution. Flow paths are build using Class VI Bioprocess-grade materials that are BSE/TSE-free and Lot-traceable.

CONFIGURED FOR YOU
Build a custom flow path to your specifications
- Engineer-designed
- Choose from over 250 components
- Connections
- Process reservoirs
- Pressure transducers
- Aseptic connectors

READY TO SHIP
Order a pre-built and quality tested off-the shelf ProConnex® Flow Path stocked for rapid delivery.
- Individually inspected
- Integrity testing available
- Clean room assembled
- Double-bagged
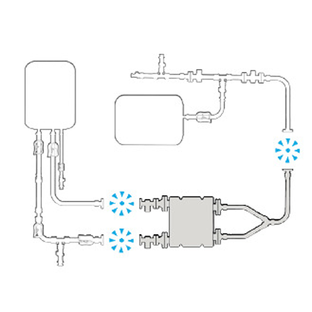
PLUG INTO EXISTING
Leverage the flexibility of genderless connectors and seamlessly connect to your existing flow path.
HIGH PERFORMANCE
Equivalent performance with ready-to-use convenience
Transition from SIUS® Cassettes to SIUS® Gamma TFF Devices seamlessly.
Membrane performance is unaffected by exposure to gamma irradiation.
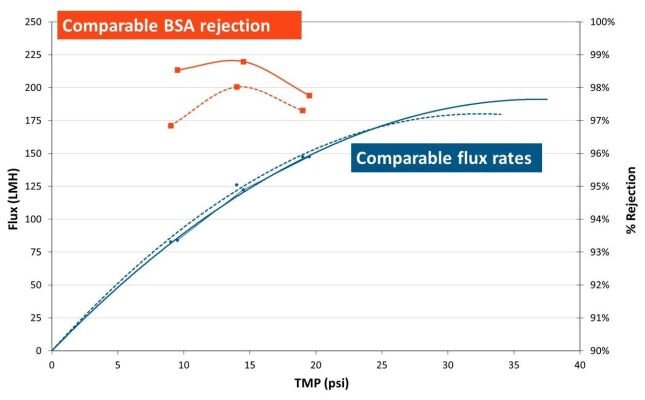
When comparing performance of TangenX® SIUS® Cassettes (solid line) and TangenX® SIUS® Gamma TFF Devices (dashed line):
- Permeate flux is comparable
- BSA rejection varies by less than 1%
HIGH PERFORMANCE
Diverse options for diverse process needs
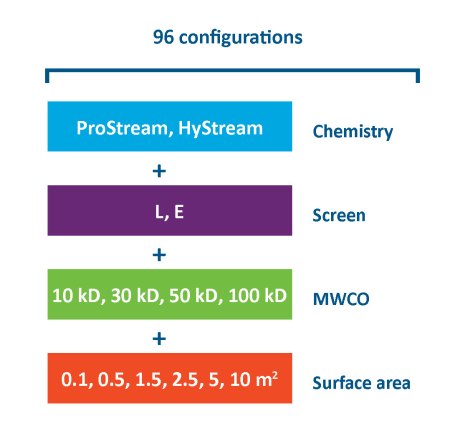
TangenX® SIUS® Gamma TFF Devices are offered with two membrane chemistries, two screen types, a wide selection of molecular weight cutoffs (MWCO), and a broad range of surface areas. With 96 configurations available, TangenX® SIUS® Gamma TFF Devices accommodate diverse process requirements for ultrafiltration and diafiltration applications from process development to commercial manufacturing.
- Neutral charge
- Low protein binding
- Excellent chemical resistance
- Hydrophilic
- Low protein binding
- minimal fouling with hydrophobic species
READY TO SCALE FAST
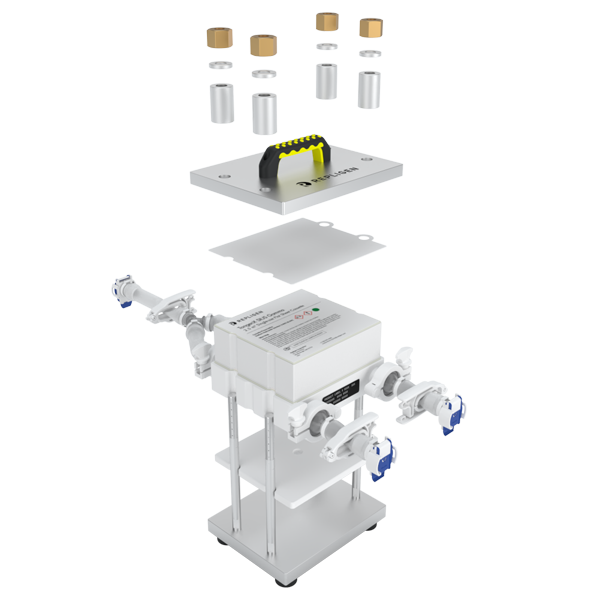
Scalable from lab to process scale
TangenX® SIUS® Gamma Devices scale from process development to large-scale manufacturing. Molecular weight cut-offs (MWCO)s extend from 10 kD up to 100 kD. Mermbrane area range from 0.1 - 10 m2.

Molecular Weight Cut-off
10 kD, 30 kD, 50 kD, 100 kD
Membrane area
0.1, 0.5, 1.5, 2.5, 5, 10 m2
TangenX® SIUS® Gamma Devices
TangenX® SIUS® Gamma TFF Devices incorporate the performance-leading TangenX® TFF membrane and cassette manufacturing technologies into a closed, gamma-irradiated, single-use assembly. Genderless, aseptic connections easily integrate into your current UF/DF flow path.
Click on the links below to explore different features.

Closed, Single-use TFF Assembly
TangenX® SIUS® Gamma TFF Devices incorporate the performance-leading TangenX® TFF membrane and cassette manufacturing technologies into a closed, gamma-irradiated, single-use assembly. Genderless, aseptic connections easily integrate into your current UF/DF flow path.
Validated manufacturing
Quality Manufacturing
TangenX® Cassettes are manufactured in a fully validated and documented manufacturing process according to the principles of cGMP. Each cassette comes with a Quality Assurance Certificate.

Comprehensive Regulatory Support Files
Each TangenX® TFF Cassette is supported by a Regulatory Support File (available upon request) that includes:
- Product information
- Cassette design
- Materials of construction
- Product performance
- Safety information
- Documentation system
- Product Manufacturing
- Qualification
- Manufacturing Process Validation
- Release Testing