Plasmid DNA Purity
SoloVPE enables quick, precise plasmid purity characterization with no dilution needed. Proprietary variable pathlength technology ensures reliable, consistent results.
Measure Plasmid Purity With Unsurpassed Accuracy
The purest assessment you can get
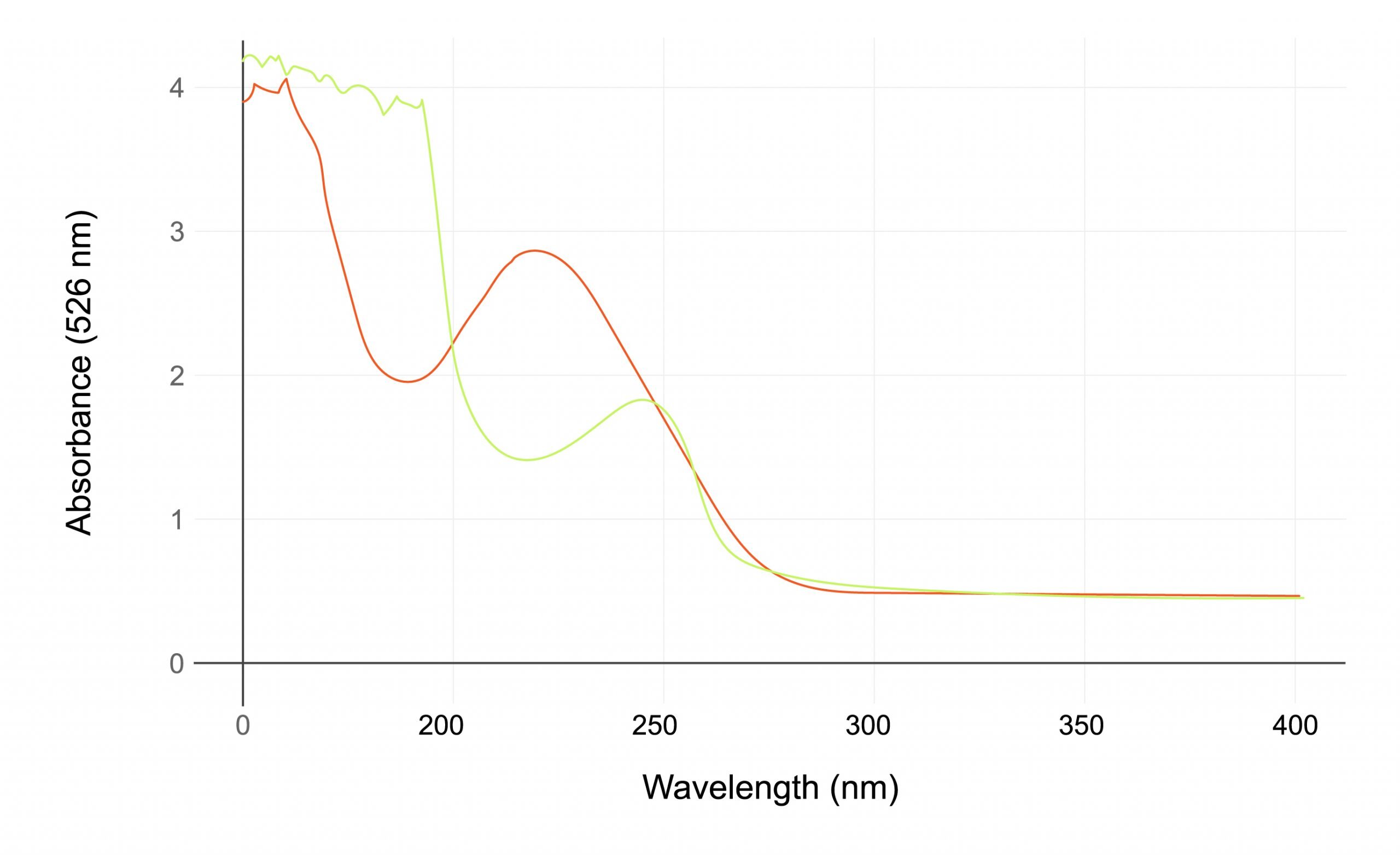
The emergence of different high absorbance biotherapeutics modalities, such as oligonucleotides, presents a challenge in using traditional spectrophotometric methods for concentration measurements. Slope Spectroscopy® offers an excellent alternative to traditional spectrophotometric analysis for nucleic acid quantitation and purity assessment using A260/A280 ratio measurement.
The SoloVPE Solution
- Accurately measures DNA and plasma purity undiluted.
- Eliminates the need for baseline and scatter correction.
- Allows users to get an accurate R2 value by measuring the 260 nm and 280 nm wavelength simultaneously.
- Manipulates Beer’s law, allowing the System to accurately vary the absorbance with the change in pathlength.
- Simultaneous wavelength measurement enables automated calculation of absorbance ratio.
- There is no sample handling or sample prep, just direct measurement, while the variable pathlength allows R2 of ≥0.999.
- The MultiQ feature allows users to measure 260 nm and 280 nm simultaneously.
Application Services
Your success is our success
CTech is committed to our customers’ success. We design service offerings to supplement our standard support options and provide you with increased access to our highly knowledgeable and experienced professionals. We offer implementation, development, and educational guidance to maximize the benefits of your variable pathlength solutions and the Slope Spectroscopy® technique.
Application Services
- 21 CFR Part 11 Guidance
- Annex 11 Guidance
- Validation Support
- Qualification Service
- Method Transfer Service
Development
- Method Design & Development
- Comparability
- On-Site Support
- Custom Standards
- Process Auditing
Education
- SOP Document Review
- Advanced Slope Spectroscopy Training
- Data Analysis
- Maintenance & Troubleshooting